a {
text-decoration: none;
color: #464feb;
}
tr th, tr td {
border: 1px solid #e6e6e6;
}
tr th {
background-color: #f5f5f5;
}
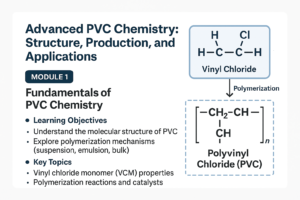
1. Molecular Structure of PVC
- PVC is a polymer derived from vinyl chloride monomer (VCM).
- Its repeating unit: –CH₂–CHCl– gives PVC its rigidity and chemical resistance.
- Chlorine atoms increase polarity, influencing mechanical and thermal properties.
2. Polymerization Mechanisms
- Suspension Polymerization:
Most common industrial method; VCM dispersed in water with initiators. - Emulsion Polymerization:
Produces fine particles for specialty applications. - Bulk Polymerization:
Less common due to heat control challenges.
3. Catalysts and Reaction Conditions
- Free-radical initiators (e.g., peroxides) start polymerization.
- Temperature and pressure control are critical for molecular weight and particle size.
Summary
PVC’s unique properties stem from its chlorine content and polymerization process. Mastering these fundamentals is key to understanding advanced formulations and applications.
Infographic Concept
Title: PVC Structure & Polymerization Overview
Elements:
- PVC repeating unit diagram.
- Flowchart of polymerization methods (Suspension → Emulsion → Bulk).
- Key initiators and conditions highlighted.